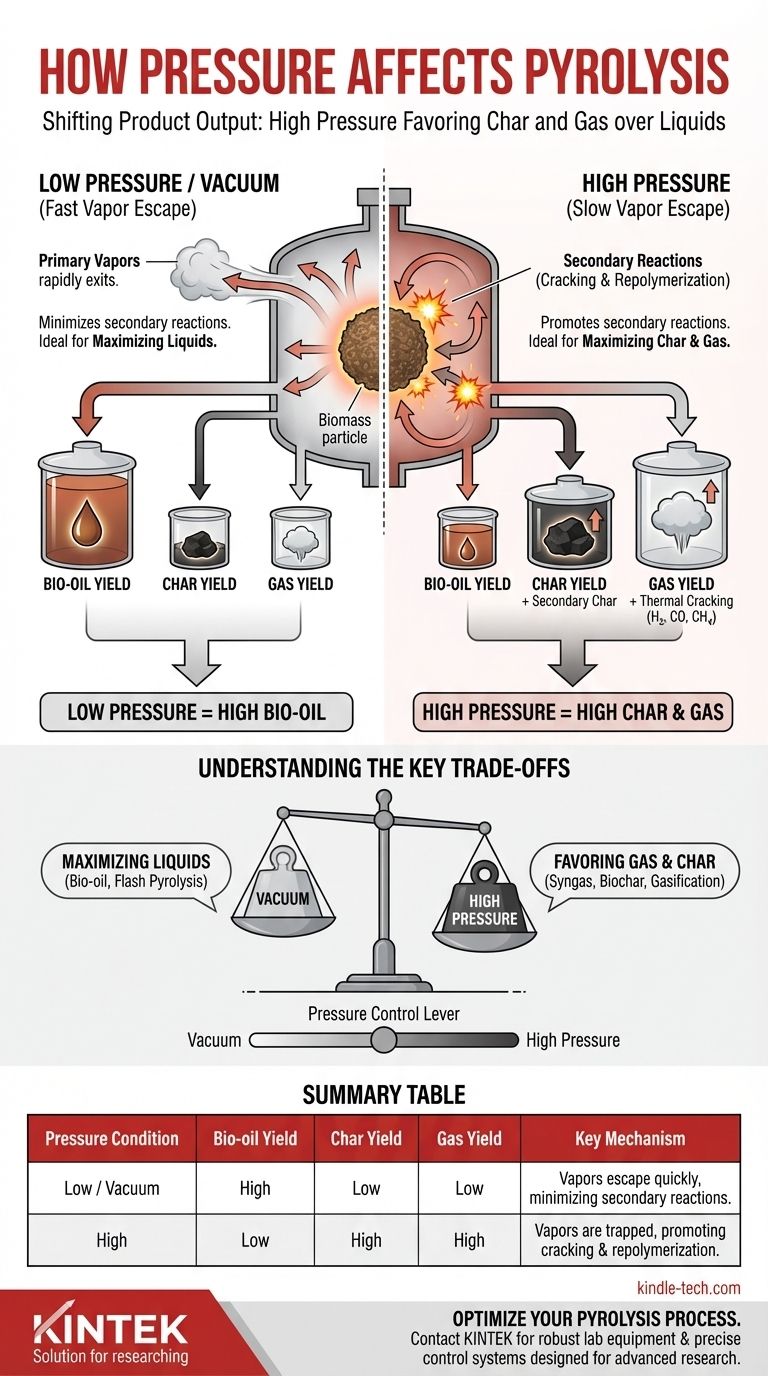
要するに、熱分解中の圧力の上昇は、製品の出力を液体からより多くの炭化物とガスへと根本的に変化させます。これは、高圧が揮発性化合物の固体バイオマスからの逃逸を物理的に妨げ、それらを二次反応が起こる高温反応ゾーンにより多くの時間を費やさせるために起こります。
圧力は熱分解における主要な制御レバーです。それは揮発性蒸気の滞留時間を直接決定し、それらがバイオオイルとして逃げるか、二次炭化物や不凝縮性ガスに変換されるかを決定します。
コアメカニズム:圧力が熱分解経路をどのように変化させるか
熱分解プロセスを制御するには、反応器内部の基本的な物理的および化学的性質を圧力がどのように変化させるかを理解する必要があります。主な影響は分子の移動にあります。
物質移動と滞留時間への影響
低圧または真空下では、揮発性化合物(蒸気)が生成されると、バイオマス粒子から逃げ出す強い駆動力があります。それらは熱い固体表面から素早く引き離されます。
高圧下では、周囲の大気がこれらの逃げ出す蒸気に押し返します。これにより、粒子および反応器からの拡散速度が大幅に遅くなり、高温ゾーンでの滞留時間が劇的に増加します。
二次反応の促進
この滞留時間の増加が、その後のすべての製品変化の根本的な原因です。一次蒸気は、熱い炭素表面の近くに閉じ込められたまま、さらなる熱分解を受けます。
これらの二次反応は、主に次の2つの経路をたどります。
- 分解(クラッキング): 蒸気が、CO、H₂、CH₄などのより小さく熱的に安定なガス分子に分解されます。
- 再重合: 蒸気が互いに、また炭素表面と反応し、二次炭化物として知られる、より安定した炭素豊富な固体に再凝縮します。

圧力の熱分解製品への影響
二次反応を制御することにより、圧力は液体、固体、ガスの最終的な収率を直接決定します。
バイオオイル(液体)収率の減少
バイオオイルは、一次熱分解蒸気を急速に冷却・凝縮させることによって生成されます。
高圧はこれらの一次蒸気がガスや炭化物の生成へと変換されるのを促進するため、反応器から出て凝縮するものが少なくなります。その結果、圧力を上げるとバイオオイル収率は体系的に減少します。
炭化物収率の増加
熱分解で生成される炭化物は、初期の固体バイオマス(一次炭化物)と蒸気の再重合(二次炭化物)の両方に由来します。
高圧は二次炭化物形成の直接的な促進要因です。これにより全体的な固体収率が高くなり、炭化物の特性が変化し、多くの場合、より高密度になります。
ガス収率の増加
閉じ込められた蒸気が不凝縮性ガスへと熱分解されるということは、高圧熱分解では常に高い量の合成ガスが生成されることを意味します。
これは、ガス化などの関連プロセスで利用される重要な原理であり、ガス生産を最大化するためにしばしば高圧下で操作されます。
主要なトレードオフの理解
運転圧力を選択することは、正しいか間違っているかの問題ではなく、特定の製品に最適化することです。選択する圧力は、液体製品と固体/ガス製品との間の根本的なトレードオフを表します。
真空熱分解:液体の最大化
真空(負圧)下での運転は、バイオオイル収率を最大化するための理想的な条件を作り出します。
真空は、蒸気が生成されるとすぐに反応器から積極的に引き出し、滞留時間を最小限に抑え、二次反応を抑制します。これはバイオ燃料生産のための「フラッシュ熱分解」の基礎となる原理です。
高圧熱分解:ガスと炭化物の優先
合成ガスまたは高収率のバイオ炭の生産が目的の場合、意図的に高圧が使用されます。
例えば、水素生産に焦点を当てたプロセスでは、蒸気の分解を最大化するために高圧を活用します。バイオ炭として炭素を隔離するように設計されたプロセスでは、有価な二次炭化物の形成を促進するために圧力が使用されます。
原料(フィードストック)の影響
原料の物理的構造は重要です。わらのような非常に多孔質な材料は、プラスチックポリマーのような高密度で非多孔質な材料よりも揮発性物質が容易に逃げ出します。
したがって、圧力の影響は、物質移動がすでに制限されている高密度の原料でより顕著になります。
目標に応じた適切な圧力の選択
運転圧力の選択は、望ましい結果を直接反映するものであるべきです。
- バイオオイル収率の最大化が主な焦点である場合: 蒸気を迅速に除去し、二次反応を防ぐために、真空下または大気圧にできるだけ近い圧力で操作します。
- 合成ガス生産の最大化が主な焦点である場合: 蒸気滞留時間を増やし、不凝縮性ガスへの熱分解を促進するために、高圧を使用します。
- バイオ炭収率の最大化が主な焦点である場合: 固体表面上での蒸気の再重合を促進するために、中程度から高圧を採用します。
結局のところ、圧力は、熱分解の化学経路を意図した製品へと導くための最も強力なツールの1つです。
要約表:
| 圧力条件 | バイオオイル収率 | 炭化物収率 | ガス収率 | 主要なメカニズム |
|---|---|---|---|---|
| 低/真空 | 高 | 低 | 低 | 蒸気が素早く逃げ、二次反応を最小限に抑える。 |
| 高 | 低 | 高 | 高 | 蒸気が閉じ込められ、分解と再重合を促進する。 |
最大の収率を得るために熱分解プロセスを最適化する準備はできましたか?
KINTEKでは、高度な熱分解の研究開発に対応した堅牢なラボ機器と消耗品の提供を専門としています。バイオオイル、合成ガス、またはバイオ炭の生産を最大化することが目標であれ、当社の反応器とシステムは、圧力などの主要なパラメータに対する正確な制御を可能にするように設計されています。
当社がお手伝いすること:
- 再現可能な結果を得るための正確な温度および圧力制御の実現。
- 特定の原料とターゲット製品に合わせた適切な機器構成の選択。
- 信頼性の高い高性能システムによるラボからパイロットスケールへのプロセスのスケールアップ。
今すぐお問い合わせいただき、お客様のアプリケーションについてご相談の上、KINTEKがお客様の熱分解の成果をどのように向上させられるかをご確認ください。お問い合わせフォームからご連絡ください – 理想的なソリューションを一緒に構築しましょう。
ビジュアルガイド